- Crescita Therapeutics (CTX) has announced excellent results from the company’s clinical trials on treating psoriasis.
- The trials tested a topical corticosteroid product, with the company’s Multiplexed Molecular Penetration Enhancer (MMPE) technology.
- The studies were conducted by Crescita partners, including Ferndale Laboratories.
- Crescita’s share price remains unchanged, with shares trading at $0.89 apiece.
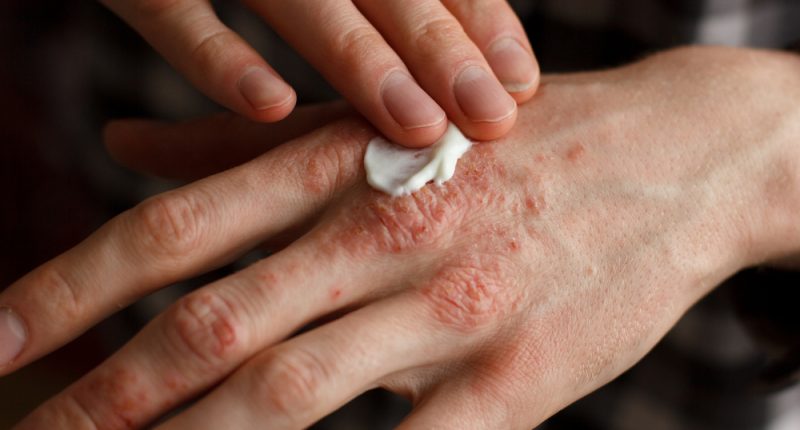
Commercial dermatology company Crescita Therapeutics (CTX) has achieved positive, topline results in 2 clinical trials on treating psoriasis.
The Phase 3 studies tested an ultra-potent topical corticosteroid currently being developed to treat plaque psoriasis. The product uses Crescita’s patented Multiplexed Molecular Penetration Enhancer (MMPE) technology.
The 2 clinical trials were conducted by partners of Crescita, including Ferndale Laboratories. The plaque psoriasis treatment is the first of 2 dermatological products being developed in collaboration with Crescita’s partners.
In both studies, a statistically significant number of patients met the parameters for successful treatment.
Crescita’s President and CEO, Serge Verreault, commented on the implications of these results. “These impressive study results support our continued belief that this product can have a clinically meaningful benefit for the treatment of patients suffering from moderate to severe plaque psoriasis.”
Under the terms of the collaboration, Crescita has used its MMPE technology to formulate 2 patented topical dermatology product candidates.
Crescita’s share price remains unchanged, with shares trading at $0.89 apiece.