- BriaCell Therapeutics (NASDAQ:BCTX) received FDA approval to begin clinical trials of Bria-BRES+, a next-generation, off-the-shelf immunotherapy for metastatic breast cancer
- The therapy is designed to activate both adaptive and innate immune systems—including T-cells, dendritic cells, and NK cells—to potentially improve effectiveness and reduce tumour immune escape
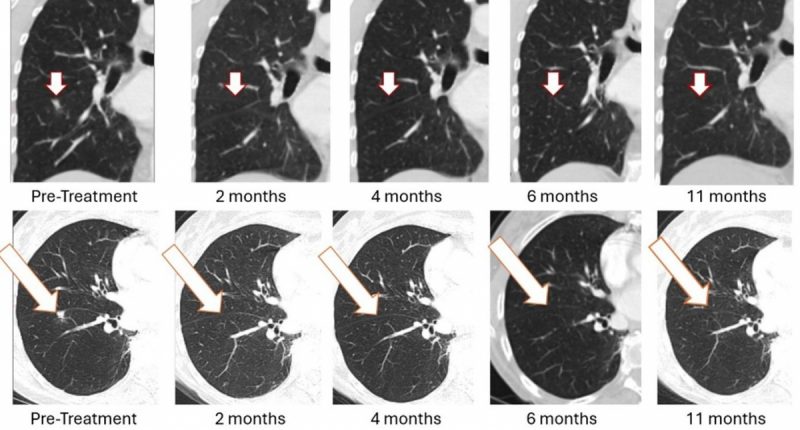
- The program builds on Bria-OTS, where one advanced breast cancer patient achieved a sustained complete response of a lung metastasis after treatment, supporting further clinical development
- BriaCell Therapeutics stock (TSX:BCT / NASDAQ:BCTX) last traded at C$5.49 / US$4.10
Clinical-stage biotechnology company BriaCell Therapeutics (TSX:BCT / NASDAQ:BCTX) has received clearance from the U.S. Food and Drug Administration (FDA) to initiate clinical evaluation of its next-generation immunotherapy candidate, Bria-BRES+. The therapy is designed as a personalized, off-the-shelf, cell-based treatment for patients with metastatic breast cancer, a population with significant unmet medical needs.
The FDA’s clearance CAN allow BriaCell to move forward with clinical testing of Bria-BRES+, which builds on the company’s existing immunotherapy platform. According to the company, the therapy is engineered to trigger a broad immune response by activating multiple components of the immune system simultaneously. This includes both adaptive and innate immune pathways, with observed activation of naïve (resting) T-cells, dendritic cells, and natural killer (NK) cells.
BriaCell believes that this multi-targeted immune engagement may offer advantages over traditional approaches by potentially improving clinical outcomes and reducing the likelihood of immune escape, a phenomenon in which tumours evade detection or destruction by the immune system. The company’s approach reflects a growing trend in oncology toward combination immune activation strategies rather than focusing on a single immune pathway.
The development of Bria-BRES+ builds on encouraging findings from BriaCell’s earlier-stage Bria-OTS™ program, which also utilizes an off-the-shelf immunotherapy approach. In that program, the company reported a notable case involving a patient with advanced metastatic breast cancer who experienced a sustained clinical response.
The patient, a 78-year-old woman with a history of multiple prior treatment failures, received Bria-OTS as a single-agent therapy. Following four doses, the patient achieved complete resolution of a lung metastasis. Imaging results initially showed a full (100%) response at approximately two months, which was subsequently confirmed at follow-up assessments at four, six, and eleven months.
The patient continued treatment, receiving a total of 17 cycles over a 12-month study period, and remains in survival follow-up, according to the company. While individual case outcomes do not establish overall efficacy, such findings are often used to inform the continued development of investigational therapies and to support further clinical evaluation.
Metastatic breast cancer, particularly in patients who have progressed after multiple lines of therapy, remains challenging to treat. Current standard therapies can offer disease control, but durable complete responses are less common, especially in heavily pretreated populations. Immunotherapy has shown promise in certain subsets of breast cancer, but broad, consistent responses have yet to be achieved across all patient groups.
BriaCell’s platform is distinguished by its use of an “off-the-shelf” model, meaning therapies are pre-manufactured and can be administered without the need for patient-specific customization at the time of treatment. This contrasts with some personalized cell therapies that require harvesting and engineering each patient’s own cells, a process that can be time-consuming and resource-intensive.
At the same time, Bria-BRES+ incorporates elements of personalization designed to improve immune recognition of tumour-specific antigens. The company has not disclosed full details of how this personalization is implemented but describes the therapy as combining scalability with individualized targeting.
Industry observers note that the balance between personalization and accessibility is a key challenge in the development of cell-based therapies. Fully individualized treatments can offer highly specific targeting but are often costly and complex to deliver, while off-the-shelf therapies may be easier to deploy but can face limitations in patient-specific efficacy. BriaCell’s approach aims to bridge that gap.
The upcoming clinical evaluation of Bria-BRES+ will be closely watched as the company seeks to demonstrate both safety and efficacy in a broader patient population. Early-phase clinical trials will likely focus on establishing dosing parameters, safety profiles, and preliminary signals of therapeutic activity.
“The unique design of Bria-BRES+ offers the potential for a favourable safety profile and meaningful therapeutic benefit in metastatic breast cancer,” BriaCell’s President and CEO, Dr. William V. Williams, said in a news release. “We look forward to advancing Bria-BRES+ into the clinic as we seek to bring new hope to these patients who have few to no effective treatment options.”
It remains to be seen whether the immune activation observed in preclinical or early clinical settings will translate into meaningful clinical benefits across a larger group of patients. However, the FDA clearance marks an important step forward for the company and highlights continued regulatory support for innovative immunotherapy approaches.
BriaCell joins a growing field of biotechnology companies exploring next-generation cancer immunotherapies, including those that target multiple aspects of the immune response. As research progresses, findings from trials such as those planned for Bria-BRES+ may help shape future treatment strategies for metastatic breast cancer.
For patients and clinicians, the advancement of new therapeutic options represents a critical area of ongoing development, particularly for those who have exhausted conventional treatments. While further study is needed, BriaCell’s latest milestone reflects continued efforts within the industry to expand the role of immunotherapy in oncology.
BriaCell is a clinical-stage biotechnology company developing cancer immunotherapies.
BriaCell Therapeutics stock (TSX:BCT / NASDAQ:BCTX) last traded at C$5.49 / US$4.10 and has lost more than 80 per cent on both exchanges since this time last year.
Join the discussion: Find out what everybody’s saying about this cancer immunotherapy stock on the BriaCell Therapeutics Corp. Bullboard and check out Stockhouse’s stock forums and message boards.