It’s a well-known and almost quaint fact that the human body is capable of regeneration. When we suffer a minor scrape or a cut, a little disinfectant, a band-aid and a few days is all it usually takes to be as good as new.
But the body’s regenerative powers are not evenly distributed. Skin, blood and certain tissues can repair themselves relatively well, while the central nervous system, including the brain, spinal cord and optic nerve, is a much more difficult frontier. Once neurons and their connections are severely damaged, the body has limited ability to rebuild them on its own, leaving patients at risk of permanent losses in movement, sensation or vision.
This article is disseminated in partnership with NurExone Biologic Inc. It is intended to inform investors and should not be taken as a recommendation or financial advice.
This is why the field of regenerative medicine is focused on ways to help the body replace or repair diseased or damaged cells through stem cells, monoclonal antibodies and even electrical stimulation. Over the past 60 years, advances such as the first bone marrow transplant in 1968 have helped catalyze this field.
That said, stem-cell-based regenerative medicine still faces major hurdles. Living-cell therapies are difficult to manufacture consistently, reproduce from batch to batch, store and scale. They may also face donor variability, immune compatibility issues, poor survival or integration after administration, and safety concerns such as unwanted differentiation or tumorigenicity in certain settings. These limitations help explain the growing interest in acellular regenerative approaches designed to capture the signaling benefits of cells without using the cells themselves.
These limitations create a significant opportunity within the US$35 billion global regenerative medicine market, which is expected to grow by more than 150 per cent to US$90 billion by 2030. There is a lucrative pathway for biotech companies to innovate and usher the field into its next era of growth.
Introducing NurExone Biologic
One company taking up this challenge head on is NurExone Biologic (TSXV:NRX)(OTC:NRXBF), market cap C$50.63 million, with ample early-stage preclinical evidence in support of its exosome-based therapies as treatments for central nervous system injuries, including spinal cord and optic nerve injuries, with treatment associated with improvements in functional and sensory recovery after a short treatment cycle.
Exosomes are cell-secreted nanoparticles that may naturally home to inflamed or damaged tissue and hold potential for wide-ranging applications in wound healing and nerve growth, including aesthetic use-cases spanning skin rejuvenation, hair growth and anti-aging in a market expected to 10x over the next decade. The benefits of exosomes versus stem cells include:
- No living cells – Exosome-based therapies are acellular, which may reduce certain safety, immune and compatibility concerns associated with cell-based therapies.
- Scalability – Easier and larger-batch production using 2D and 3D reactors, both of which are part of NurExone’s production process.
- Storage and handling – Exosomes can potentially be frozen and thawed without losing effectiveness. This may simplify storage, handling and supply-chain requirements compared with living stem cells.
- Minimally invasive administration -Given exosomes’ affinity for distressed tissue, they support less invasive treatment approaches.
NurExone is developing its proprietary ExoTherapy platform to capitalize on exosomes’ healing and regenerative properties, deploying in-house capabilities to produce the nanoparticles, synthesize therapeutic cargo, load that cargo onto the nanoparticles and develop them into nanodrugs for testing and potential commercialization. The company has a handful of strategic unmet needs in its sights, each of which is tied to a multi-billion-dollar market.
ExoPTEN for acute spinal cord injuries
NurExone’s first nanodrug, ExoPTEN, uses as its cargo a proprietary RNA sequence to inhibit the expression of the PTEN tumor-suppressor gene, commonly found in neurons and regenerating axons, with numerous studies showing that PTEN inhibition can promote wound repair and nerve growth after injury.
ExoPTEN’s first targeted use-case is the regeneration of spinal neurons, vying to improve quality of life for the more than 900,000 new traumatic spinal cord injury patients per year, representing a US$7.9 billion opportunity to ease the strain on the global healthcare system.
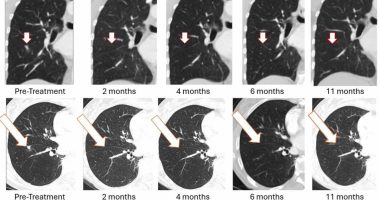
The drug is the first known minimally invasive therapy shown to foster nerve regeneration following a full spinal cord transection in preclinical studies. About 75 per cent of animals in the study recovered motor function and results were replicated across multiple experiments in NurExone’s labs. While monoclonal antibodies and epidural electrical stimulation can be developed into non-immunogenic, off-the-shelf products, they have not demonstrated the ability to repair a full spinal cord transection, making ExoPTEN a comparatively better candidate for a new standard of care in the regenerative medicine landscape.
ExoPTEN’s promising, early-stage evidence led the US Food and Drug Administration and the European Medicines Agency to award it the Orphan Drug Designation for acute spinal cord injuries, granting NurExone potential benefits including fee reductions, an expedited clinical process, as well as 7-12 years of market protection, reinforcing the company’s pathway to clinical trials in the US and Europe.
With millions of people with spinal cord injuries standing to benefit from better treatment options, NurExone is pursuing Investigational New Drug status with the US FDA, keen to kick off phase-1 human trials in 1H 2027 and further demonstrate how its stock price is a poor reflection of the ExoTherapy platform’s clinical and commercial potential.
ExoPTEN for acute glaucoma
NurExone is also studying ExoPTEN as a therapy to treat acute glaucoma, a disease that damages the optic nerve, putting vision at risk for 350,000-700,000 new people every year. With vision loss and blindness costing the US about US$26,900 per patient annually, this equates to a more than US$9 billion opportunity, robustly incentivizing innovators like NurExone to step in and solve this global health burden.
The company has also delivered successful pre-clinical results under this indication, substantiating optic nerve healing and vision restoration in 75 per cent of animals in a study.
A larger-scale pre-clinical study is currently underway at the Sheba Medical Center, ranked as the 8th best hospital in the world by Newsweek in 2025, which will support a phase-I clinical trial envisioned for 1H 2027.
NurExone leadership has taken its future into its own hands by establishing a US subsidiary to anchor a push into the North American market.
Exo-Top
Exo-Top, NurExone’s wholly owned US subsidiary, will lead the company’s North American manufacturing, development and commercialization strategy, harnessing a Master Cell Bank of GMP-grade human bone marrow mesenchymal stem cells (MSC) as a foundation for exosome production, ensuring consistency during the transition from the preclinical to the commercial realm.
The US subsidiary, focused on production and supply, will work in close proximity with NurExone Biologic Ltd., tasked with R&D and product development, leveraging a sublicense agreement that gives it biomanufacturing rights to the technology underlying the ExoTherapy platform.
With the freedom to grow its business in the world’s top economy, Exo-Top has wasted no time solidifying its presence, recently signing a manufacturing letter of intent (LOI) with Florida-based BioXtek to explore MSC exosome production domestically and abroad for a variety of potential use-cases, including wound care, pain management and orthopedics.
BioXtek, a clinical-stage company specializing in research, clinical trials and GMP manufacturing of exosomes and other biological products, brings established commercial relationships and an 11,000-square-foot production facility to the potential deal, including proven quality control systems to ensure consistent, medical-grade products.
Combining Exo-Top’s Master Cell Bank with BioXtek’s manufacturing capabilities, the companies plan to accelerate ExoPTEN’s clinical timeline and address exosome demand across the field of regenerative medicine.
The LOI, subject to a definitive agreement, could see Exo-Top generate initial revenue as soon as 1H 2027.
NurExone’s versatile leadership team
NurExone’s go-to-market strategy is being catalyzed by an executive team with proven expertise across its target markets, with foundations in science to excel in the lab, finances to raise capital and business development to turn innovation into shareholder value. Here’s a quick look at key members and what they bring to the company:
- Director and Chief Executive Officer, Dr. Lior Shaltiel, has worked for nano-drug delivery companies, including LipoCure and Ayana Pharma, and previously served as Partner and Vice President at a Chinese investment bank, where he evaluated opportunities in the Israeli pharmaceutical industry.
- Chairman, Co-Founder and VP of Strategic Development, Yoram Drucker, has been building businesses in the Israeli biotech industry for more than 20 years, including the co-founding of technology companies Pluri (NASDAQ:PLUR), Brainstorm (USOTC:BCLI) and InnoCan (CSE:INNO).
- Chief Financial Officer, Eran Ovadya, has built a more than two-decade career in corporate finance, accounting, M&A, IPOs and RTOs. He oversaw finances for numerous biotech companies, including Gamida Cell, West Pharma Israel, Leap Therapeutics and Omrix Biopharmaceuticals, a division of Ethicon Biosurgery, a Johnson & Johnson company.
- R&D Director, Dr. Tali Kizhner, has been developing therapeutic proteins and dietary supplements for more than 15 years, with a specialty in the intracellular delivery of biological payloads. She previously worked for International Fragrance and Flavors and Protalix Biotherapeutics, where she contributed to EMEA and FDA-approved products.
- Head of CMC, Quality and Regulation, Dr. Ina Sarel, brings more than 20 years of biotechnology product development experience from discovery to clinical trials, including stem/progenitor cell therapy.
- Head of Operations, Ram Petter, is a 28-year veteran within the biotech and biopharmaceutical fields, specializing in early development, process optimization, GMP manufacturing, partnerships and business development, with a focus on recombinant proteins and monoclonal antibody-based products.

These highly-accomplished executives, supported by well-rounded researchers and world-class scientific advisors, are setting NurExone on course to usher its exosome drug candidates along the clinical development cycle, pursue licensing deals for the ExoTherapy platform and further establish the company as a near-term leader in regenerative medicine.
This potentially value-accretive trajectory begs the question about what NurExone’s investment prospects look like today.
A potential market leader at a cut-rate price
Exosomes’ stronger therapeutic potential versus stem cell therapy, combined with promising pre-clinical data in support of lead candidate ExoPTEN for spinal cord injuries and acute glaucoma, make NurExone an early-stage contender for revolutionizing and catalyzing development in regenerative medicine, which, as we have seen, afford it a more than US$16 billion addressable market.
When we compare this figure to NurExone’s current market cap of C$50.63 million, we notice that the company is being valued at only 0.23 per cent of its addressable market, which is an excessively conservative appraisal, given ExoPTEN’s positive early-stage milestones towards improving the quality of life for millions of people living with central nervous system injuries.
As clinical trials get underway, a reasonable investor should expect the market to rapidly come to its senses as new milestones cross the wire, closing the gap between NurExone’s stock price and the underlying company’s potential to shape the future of regenerative medicine. This shift in market sentiment would, in turn, better equip leadership – strongly aligned with shareholders at 25 per cent founder ownership – to optimize growth initiatives, reinforce revenue streams and ultimately scale into profitability.
Join the discussion: Find out what investors are saying about this micro-cap healthcare stock on the NurExone Biologic Inc. Bullboard and make sure to explore the rest of Stockhouse’s stock forums and message boards.