- NuGen Medical Devices (NGMD) announced that it intends to complete a non-brokered private placement of up to $3,600,000
- NuGen will use the gross proceeds for working capital and for general corporate purposes
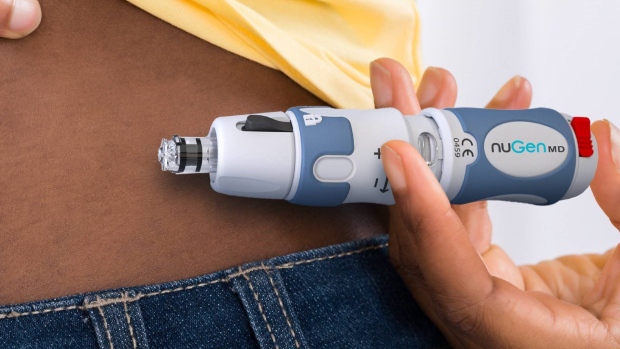
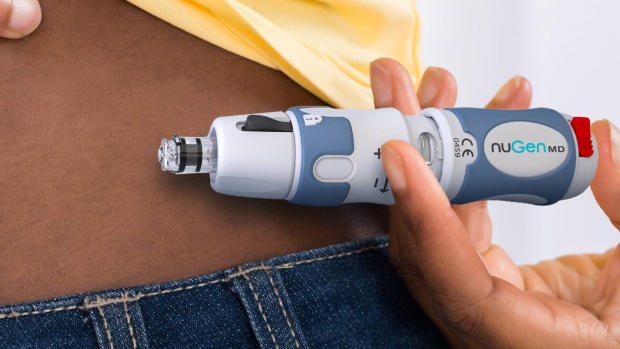
- NuGen is an emerging specialty medical device company developing the next generation of novel drug delivery technologies
- Nugen Medical Devices Inc. (NGMD) is holding steady, trading at C$0.22 per share at 2:20 pm ET
NuGen Medical Devices (NGMD) announced that it intends to complete a non-brokered private placement of up to $3,600,000.
The company plans to issue up to 20,000,000 units at $0.18 per unit. Each unit consists of one common share and one common share purchase warrant. Each warrant entitles the holder to purchase one additional common share at $0.24 for twenty-four months from the closing date. The warrants are subject to the acceleration clause.
NuGen will use the gross proceeds for working capital and for general corporate purposes.
The company may pay finders fees consisting of 8 per cent cash and 8 per cent broker warrants. Each broker warrant entitles the holder to acquire one unit at $0.18 for two years from the date of issuance.
All securities issued will be subject to a statutory hold period of four months. Closing of the offering is subject to receipt of all necessary corporate and regulatory approvals.
About NuGen Medical Devices
NuGen is an emerging specialty medical device company developing the next generation of novel drug delivery technologies. Its principal business is the research, development and commercialization of innovative needle-free injection devices and systems for the administration of subcutaneous medication.
Nugen Medical Devices Inc. (NGMD) is holding steady, trading at C$0.22 per share at 2:20 pm ET.