- Scientists created the first‑ever molecule with a half‑Möbius electronic topology, a configuration never previously observed or predicted
- The molecule’s unusual corkscrew‑like electron motion was revealed using IBM’s (NYSE:IBM) quantum‑centric supercomputing
- The discovery shows that electronic topology can be engineered, marking a major advance for both chemistry and quantum computing
- IBM stock (NYSE:IBM) opened trading at US$256.44
An international team of researchers from IBM (NYSE:IBM), The University of Manchester, Oxford University, ETH Zurich, EPFL, and the University of Regensburg has synthesized a molecule unlike anything previously observed in chemistry — and used quantum computing to understand it.
The discovery, published in Science, marks the first experimental observation of a half‑Möbius electronic topology in a single molecule, a configuration long theorized but never realized.
The breakthrough offers a rare dual advancement: it introduces a completely new class of molecular electronic behaviour while demonstrating quantum computing’s emerging power as a scientific tool capable of modelling systems too complex for classical computers.
“First, we designed a molecule we thought could be created, then we built it, and then we validated it and its exotic properties with a quantum computer,” Alessandro Curioni, IBM fellow, vice president, Europe and Africa, and director of IBM Research Zurich, explained in a media statement. “This is a leap towards the dream laid out by renowned physicist Richard Feynman decades ago to build a computer that can best simulate quantum physics and a demonstration where, as he said, ‘There’s plenty of room at the bottom.’ The success of this research signals a step towards this vision, opening the door for new ways to explore our world and the matter within it.”
A molecule with a twist — literally
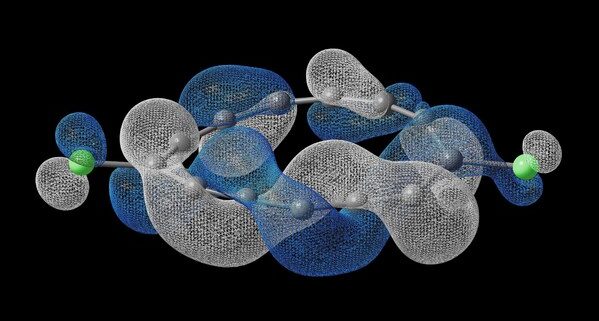
The molecule, designated C₁₃Cl₂, was built atom‑by‑atom at IBM using a custom precursor supplied by Oxford University. Under ultra‑high vacuum and temperatures near absolute zero, IBM scientists used finely tuned voltage pulses to remove individual atoms with precision.
Scanning tunnelling and atomic force microscopy — technologies pioneered at IBM — revealed something unprecedented: the molecule’s electrons traverse its structure with a corkscrew‑like motion, completing a 90‑degree twist with each cycle. Unlike familiar Möbius structures, which require a full 360‑degree twist, this “half‑Möbius” state takes four loops to return to its starting phase.
Even more striking, the molecule’s electronic topology is reversible. Under controlled conditions, researchers switched it between clockwise‑twisted, counterclockwise‑twisted, and untwisted states. The finding suggests that electronic topology can be engineered on demand, rather than discovered passively in nature.
Where classical computers fail, quantum systems step in
Understanding why this exotic topology formed posed a major computational challenge. Electron behavior inside C₁₃Cl₂ is heavily entangled — each electron’s behavior depends on the simultaneous configuration of all others. Classical computers struggle with such exponential complexity.
Quantum computers, however, operate under the same quantum rules that govern molecular electrons. Leveraging IBM’s quantum systems within a quantum‑centric supercomputing workflow, the research team simulated the molecule’s electronic structure with high fidelity.
The simulation uncovered helical molecular orbitals characteristic of the half‑Möbius topology and revealed the underlying mechanism: a helical pseudo–Jahn-Teller effect, a phenomenon that alters molecular geometry through quantum-level interactions.
This marks one of the clearest real‑world demonstrations to date of quantum computing providing scientific insight unattainable by classical methods.
A milestone in quantum‑centric supercomputing
The project showcases the emerging model of hybrid computing that IBM has been promoting: quantum‑centric supercomputing, which coordinates CPUs, GPUs, and quantum processing units to tackle distinct parts of a scientific problem.
By using each component where it excels, researchers overcame computational barriers that would have stalled conventional approaches.
Building on IBM’s legacy in nanoscale innovation
This discovery continues IBM’s long history of pushing the boundaries of atomic‑scale research. The company invented the scanning tunnelling microscope in 1981 — work that earned a Nobel Prize — and in 1989 achieved the first deliberate manipulation of individual atoms.
Over the decades, IBM’s teams have used these methods to construct increasingly complex molecular structures. With today’s announcement, they have crossed into new territory: creating a molecule with no precedent in nature or theory, and revealing its behaviour with the help of quantum computation.
A new frontier for chemistry and quantum science
The creation of this molecule and the understanding of its half‑Möbius topology suggest a future in which chemists can design electronic behavior at will, and quantum computers play an essential role in validating and exploring these frontier systems.
As the research team notes, this is likely only the beginning. With quantum‑centric workflows now capable of tackling problems beyond the reach of classical machines, scientists may soon uncover — or create — entirely new realms of molecular behaviour.
About IBM
International Business Machines Corp. is a global hybrid cloud, AI and consulting company with clients in more than 175 countries.
IBM stock (NYSE:IBM) opened 0.11 per cent higher at US$256.44 and has risen more than 3 per cent since this time last year.
Join the discussion: Find out what the Bullboards are saying about IBM and check out Stockhouse’s stock forums and message boards.
Stockhouse does not provide investment advice or recommendations. All investment decisions should be made based on your own research and consultation with a registered investment professional. The issuer is solely responsible for the accuracy of the information contained herein. For full disclaimer information, please click here.